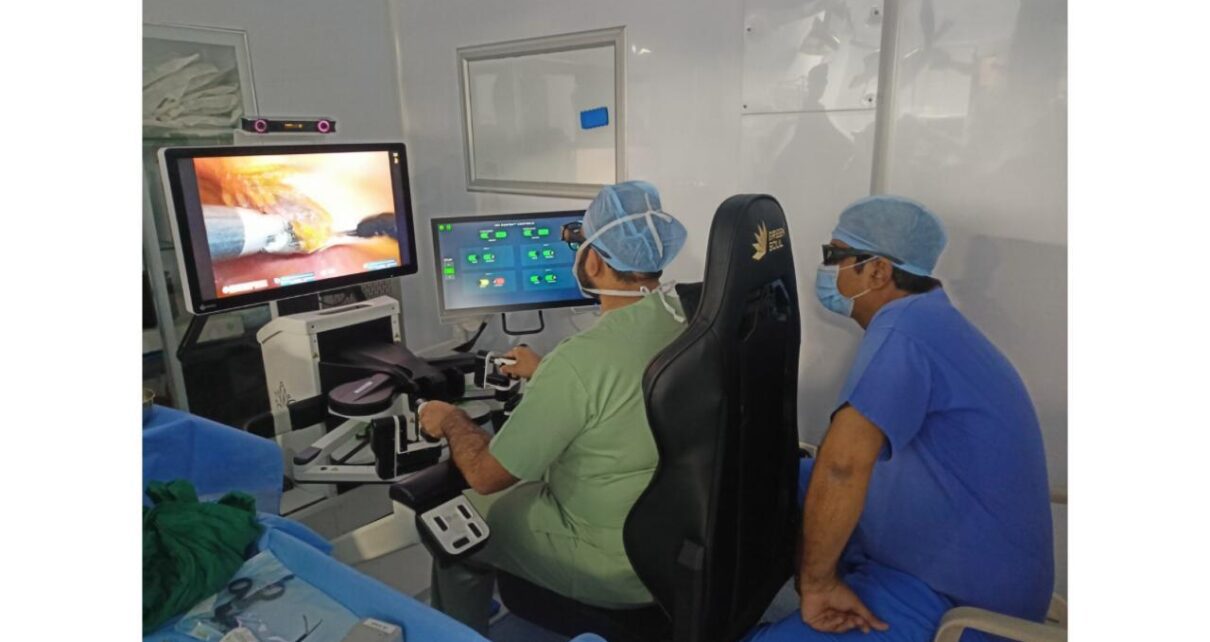
CMJ Hospital, a beacon of healthcare for the poor & needy in Girgaon, Mumbai, with a legacy spanning 60+ years, has announced a groundbreaking advancement in surgical care with the introduction of robotic surgery on the SSI Mantra platform. This initiative is aligned with CMJ Hospital’s commitment to providing world-class treatment to the community, particularly […]
Tag: Medical Innovation
IlluminOss Subsidiary ABL Medical Developing Anti-Microbial Blue Light Fiber Technology!
ABL Medical Inc. a subsidiary of IlluminOss Medical Inc. In 2018, IlluminOss Medical began offering a system in the US that used blue light delivered via a long fiber-optic catheter to harden light-curing liquid within balloon-like orthopedic implants. Now, data from ABL Medical demonstrates that blue light with fiber-optic technology may be adapted for antimicrobial […]
Kane Biotech Files New Patent on revyve™ Antimicrobial Wound Gel Spray
Kane Biotech Inc. (TSX-V:KNE OTCQB:KNBIF) (the “Company” or “Kane Biotech” or “Kane”) announces that it has filed a patent on its revyve™ Antimicrobial Wound Gel Spray, a follow-on product to its FDA 510(k) cleared revyve™ Antimicrobial Wound Gel, and will be introducing it today at the Boswick Burn and Wound Care Symposium. “Kane has taken […]
YASHODA HOSPITALS, Hyderabad SUCCESSFULLY TREATS ELDERLY MAN WITH CA. BUCCAL MUCOSA INFILTRATING INTO MANDIBLE
Yashoda Hospitals (Malakpet) Hyderabad, a leading healthcare institution, has successfully treated an elderly man aged 57 from Warrangal suffering from CA. Buccal Mucosa, commonly known as oral cancer. The patient was first admitted under Dr. Hari Krishna Reddy. K, Consultant General & Laparoscopic Surgeon, Yashoda Hospitals Hyderabad, who then referred it to Dr. Shaik Saleem, […]
Inspira™ Technologies Signs Letter of Intent with Northwestern Memorial Hospital in Chicago for Clinical Use of INSPIRA™ ART100 Device
Inspira Technologies OXY B.H.N Ltd. (Nasdaq: IINN) (Nasdaq: IINNW) (the “Company” or “Inspira Technologies”), a pioneer in life support technology with a vision to supersede traditional mechanical ventilators, has announced the signing of a non-binding Letter of Intent (LOI) with Northwestern Memorial Hospital in Chicago, Illinois (“Northwestern”). Northwestern, ranked as the eighth best hospital in the United States by Newsweek in 2023, is part of […]
Citius Pharmaceuticals Completes Enrollment in Pivotal Phase 3 Trial of its Mino-Lok® Therapeutic to Salvage Catheters
Citius Pharmaceuticals, Inc. (“Citius” or the “Company”) (Nasdaq: CTXR), a late-stage biopharmaceutical company dedicated to the development and commercialization of first-in-class critical care products, today announced that it has completed enrollment in its pivotal Phase 3 clinical trial for Mino-Lok®, an antibiotic lock solution to salvage catheters in patients with catheter-related bloodstream infections. A total of […]
Ocutrx Unveils OcuLenz AR/XR Headset, Aiding Vision for Patients with Advanced Macular Degeneration
Ocutrx Technologies, Inc., a leader in augmented reality (AR) and 3D technology for medical and surgical applications, today is pulling back the curtain on its latest innovation: OcuLenz™, its groundbreaking AR/XR headset. OcuLenz is engineered to enhance visual clarity for individuals living with advanced macular degeneration (AMD). This innovative AR/XR headset is designed to be lightweight […]
Vesica Health Appoints Distinguished Urologic Oncologist Dr. William T. Lowrance to Chief Medical Officer
VESICA HEALTH, INC., a multi-omics, liquid biopsy company transforming the management of hematuria patients and improving the early detection of bladder cancer, today announced that William T. Lowrance, MD, MPH, MBA will serve as the company’s Chief Medical Officer. Dr. Lowrance brings more than 20 years of clinical experience, as a professor of urology and a […]
First Results with Erdafitinib-Releasing Intravesical Delivery System (TAR-210) Show Early Evidence of Positive Clinical Activity in Patients with Non-Muscle-Invasive Bladder Cancer with Select Fibroblast Growth Factor Receptor Alterations
The Janssen Pharmaceutical Companies of Johnson & Johnson announced today the first results from an open-label, multicenter Phase 1 study evaluating the safety and efficacy of TAR-210, an intravesical delivery system designed to provide sustained, local release of erdafitinib into the bladder in patients with non-muscle-invasive bladder cancer (NMIBC) with select fibroblast growth factor receptor (FGFR) […]
AMGEN PRESENTS NEW LUMAKRAS® (SOTORASIB) PLUS VECTIBIX® (PANITUMUMAB) DATA IN PATIENTS WITH KRAS G12C-MUTATED METASTATIC COLORECTAL CANCER
Amgen (NASDAQ:AMGN) today announced data from the global Phase 3 CodeBreaK 300 trial evaluating two doses of LUMAKRAS® (sotorasib) (960 mg or 240 mg) in combination with Vectibix® (panitumumab). Both doses demonstrated a statistically significant superiority in progression-free survival (PFS) over the investigator’s choice of therapy in patients with chemorefractory KRAS G12C-mutated metastatic colorectal cancer (mCRC). The results are being presented today […]